Online Database of Chemicals from Around the World
| Beijing Huarui Union Bio-med Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bjhrunion.com | |||
 | +86 (10) 5975-3109 | |||
 | info@bjhrunion.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Carbides and carbonates |
|---|---|
| Name | Lanthanum carbonate octahydrate |
| Molecular Structure | 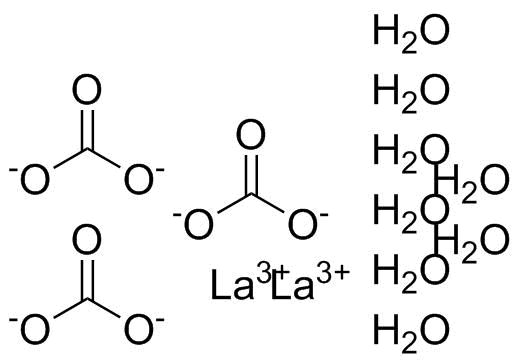 |
| Molecular Formula | La2(CO3)3.8H2O |
| Molecular Weight | 601.96 |
| CAS Registry Number | 6487-39-4 |
| EC Number | 691-114-5 |
| SMILES | C(=O)([O-])[O-].C(=O)([O-])[O-].C(=O)([O-])[O-].O.O.O.O.O.O.O.O.[La+3].[La+3] |
| Density | 2.6 - 2.7 g/mL (Expl.) |
|---|---|
| SDS | Available |
|---|---|
|
Lanthanum carbonate octahydrate is an inorganic coordination compound with the chemical formula La2(CO3)3·8H2O. It consists of lanthanum(III) cations (La3+), carbonate anions (CO32−), and water molecules of crystallization. The compound belongs to the class of rare earth metal carbonates and typically forms hydrated crystalline solids with low solubility in water. The structure is based on La3+ ions coordinated by oxygen atoms from carbonate groups and water molecules. The carbonate ion has a trigonal planar geometry, with delocalized bonding over the three oxygen atoms. In the solid state, lanthanum carbonate octahydrate forms an extended ionic lattice stabilized by electrostatic interactions between the large trivalent lanthanum ions and the doubly charged carbonate ions, along with hydrogen bonding involving the coordinated water molecules. Lanthanum carbonate compounds were first studied as part of broader investigations into rare earth element chemistry in the 19th and early 20th centuries, following the isolation and characterization of lanthanum from mineral sources. Systematic study of lanthanide carbonates helped establish trends in ionic radii, hydration behavior, and coordination chemistry across the rare earth series. Lanthanum carbonate octahydrate is typically prepared by reacting soluble lanthanum salts, such as lanthanum chloride or lanthanum nitrate, with a source of carbonate ions, such as sodium carbonate or ammonium carbonate, in aqueous solution. The low solubility of lanthanum carbonate leads to precipitation of the hydrated solid, which can incorporate water molecules into its crystal lattice during formation. In aqueous environments, lanthanum carbonate has very low solubility due to the strong lattice energy of the lanthanum–carbonate system. When it comes into contact with acidic conditions, it reacts to release lanthanum ions and carbon dioxide gas, a typical behavior of metal carbonates. This acid–base reactivity is important in its pharmaceutical application. A key application of lanthanum carbonate octahydrate is in medicine, where it is used as a phosphate binder for the treatment of hyperphosphatemia in patients with chronic kidney disease. In the gastrointestinal tract, lanthanum ions released from the carbonate react with dietary phosphate to form insoluble lanthanum phosphate, which is then excreted in the feces. This reduces phosphate absorption into the bloodstream and helps control serum phosphate levels. The mechanism of action is based on the strong affinity of La3+ for phosphate ions (PO43−). Lanthanum phosphate is highly insoluble, and its formation effectively sequesters phosphate in the digestive tract. Because the compound acts locally in the gut and has minimal systemic absorption, it is suitable for long-term management of phosphate balance. Pharmacologically, lanthanum carbonate octahydrate is administered orally in chewable or tablet form. It dissociates in the acidic environment of the stomach, releasing lanthanum ions that bind phosphate. The compound is designed to remain largely unabsorbed, with most of the lanthanum excreted in the feces. From a materials chemistry perspective, lanthanum carbonate octahydrate reflects typical properties of lanthanide carbonates, including low solubility, strong ionic interactions, and extensive hydration. The presence of eight water molecules in the crystal structure contributes to its stability and physical characteristics. Overall, lanthanum carbonate octahydrate is a hydrated rare earth carbonate with important medical application as a phosphate-binding agent in chronic kidney disease. Its chemistry is governed by the strong affinity between lanthanum ions and carbonate and phosphate anions, and its low solubility underlies both its solid-state stability and its therapeutic function. References 2025. Control of phosphorus release from sediment by lanthanum carbonate-modified bentonite (LCBT) alone and in combination with Myriophyllum aquaticum: comparison of LCBT with lanthanum-modified bentonite and iron/aluminum co-modified calcite, and efficiency, mechanisms, and environmental safety of LCBT-Myriophyllum aquaticum combination. Journal of Soils and Sediments. DOI: 10.1007/s11368-025-04137-0 2020. Suppression of phosphorus release from sediment using lanthanum carbonate as amendment. Environmental science and pollution research international. DOI: 10.1007/s11356-020-10714-2 |
| Market Analysis Reports |