Online Database of Chemicals from Around the World
| Chemtrade International Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.trade-chem.com | |||
 | +86 (532) 8689-3005 | |||
 | +86 (532) 8689-3005 | |||
 | chemtrade@trade-chem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Openchem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.openchem.com.cn | |||
 | +86 (21) 5028-0880 | |||
 | +86 (21) 5028-0883 | |||
 | info@openchem.com.cn | |||
| Chemical manufacturer since 1988 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ningbo EST Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.estchemical.com | |||
 | +86 18868651166 | |||
 | +86 (574) 5670-2932 | |||
 | sales@estchemical.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Guangzhou Jun Xin Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxinchem.com | |||
 | +86 (020) 8256-9208 | |||
 | +86 (020) 2826-9100 | |||
 | ivyyoung@gzjxchem.com | |||
 | QQ Chat | |||
 | WeChat: gzjx18122041046 | |||
 | WhatsApp:+8618122041046 | |||
| Chemical distributor since 2007 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Nantong Acetic Acid Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ntacf.com | |||
 | +86 (513) 8554-5538 | |||
 | +86 (513) 8554-4224 | |||
 | gord@ntacf.com | |||
| Chemical manufacturer since 1959 | ||||
| Classification | Surfactant >> Anionic surfactant >> Sulfate type |
|---|---|
| Name | Ammonium lauryl sulfate |
| Synonyms | Ammonium dodecyl sulphate; Dodecyl sulfate ammonium salt |
| Molecular Structure | 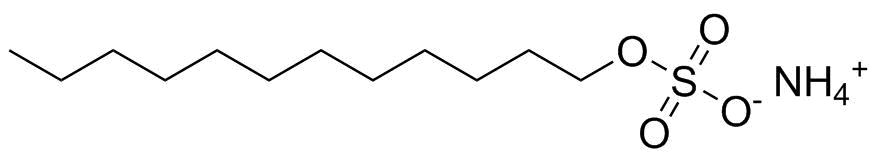 |
| Molecular Formula | C12H25O4S.NH4 |
| Molecular Weight | 283.42 |
| CAS Registry Number | 2235-54-3 |
| EC Number | 218-793-9 |
| SMILES | CCCCCCCCCCCCOS(=O)(=O)[O-].[NH4+] |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H318-H319 Details | ||||||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P302+P352-P305+P351+P338-P305+P354+P338-P317-P321-P330-P332+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Ammonium lauryl sulfate is an anionic surfactant commonly used in detergents, shampoos, and personal care formulations. It is the ammonium salt of lauryl sulfuric acid (dodecyl sulfate), with the chemical structure consisting of a 12-carbon linear alkyl chain (lauryl group) attached to a sulfate ester group, neutralized by an ammonium cation (NH4+). Its amphiphilic nature, combining a hydrophobic hydrocarbon chain and a hydrophilic sulfate head group, gives it strong surface-active properties. The compound belongs to the class of alkyl sulfate surfactants, which are widely used in cleansing and emulsifying applications. These surfactants reduce surface tension between water and oils, allowing the formation of micelles that can solubilize hydrophobic substances such as sebum, grease, and particulate dirt. This property makes ammonium lauryl sulfate effective as a foaming and cleansing agent. The history of alkyl sulfate surfactants is closely linked to the development of synthetic detergents in the early 20th century. As natural soap production based on fatty acid salts reached its limitations in hard water conditions, researchers developed sulfate-based surfactants derived from fatty alcohols. Lauryl sulfate derivatives became particularly important due to their strong cleaning performance and ability to produce stable foam. Ammonium lauryl sulfate is typically produced through the sulfation of lauryl alcohol (dodecanol), followed by neutralization with ammonia. In the first step, lauryl alcohol reacts with sulfur trioxide or chlorosulfonic acid to form lauryl sulfuric acid or its intermediate sulfate ester. This is then neutralized with ammonia to produce the ammonium salt. The resulting compound is often supplied as an aqueous solution or paste due to its hygroscopic nature. In aqueous solution, ammonium lauryl sulfate dissociates into ammonium ions and lauryl sulfate anions. The lauryl sulfate anion is responsible for its surfactant properties. At concentrations above the critical micelle concentration, the molecules self-assemble into micelles, with hydrophobic tails oriented inward and hydrophilic sulfate groups facing the aqueous environment. These micelles enable emulsification and removal of oily substances. Ammonium lauryl sulfate is widely used in personal care products such as shampoos, body washes, facial cleansers, and toothpaste. It is valued for its strong foaming ability and effective cleansing performance. It is often used in combination with other surfactants, including amphoteric and nonionic surfactants, to adjust viscosity, mildness, and foam characteristics in formulations. Compared with sodium lauryl sulfate, ammonium lauryl sulfate is sometimes considered slightly milder in certain formulations, although both are strong anionic surfactants. The choice of counterion (ammonium versus sodium) can influence properties such as solubility, foam texture, and formulation compatibility. In industrial applications, ammonium lauryl sulfate is also used in household cleaning products, liquid detergents, and industrial degreasers. Its ability to remove oils and particulates makes it effective in a wide range of cleaning systems. From a physicochemical perspective, the surfactant properties of ammonium lauryl sulfate arise from its amphiphilic structure and its ability to reduce interfacial tension. The long alkyl chain provides hydrophobic interactions, while the sulfate head group interacts strongly with water through ion–dipole interactions and hydrogen bonding. Overall, ammonium lauryl sulfate is a widely used anionic surfactant derived from lauryl alcohol. Its combination of strong cleansing ability, foaming properties, and formulation versatility has made it an important ingredient in personal care and cleaning products. Its development is part of the broader history of synthetic surfactants designed to improve upon traditional soap systems. References 2026. Classification of industrial chemicals for respiratory chemosensory irritation using the TRPV1-expressing neuronal SH-SY5Y cell model and machine learning. Archives of Toxicology. DOI: 10.1007/s00204-025-04288-6 2026. A Mini Review on Surfactants: Production, Applications, Recent Advances, and a Way Toward Biosurfactant. Journal of Solution Chemistry. DOI: 10.1007/s10953-026-01562-8 2025. Applications of Surfactants in Personal Care Products. Industrial Applications of Individual and Mixed Surfactant Systems - Volume 1. DOI: 10.1007/978-981-95-4609-1_4 |
| Market Analysis Reports |